Researchers have detected, for the first time, a complex organic chiral molecule outside of our solar system, paving the way to understanding one of the most puzzling mysteries of the early origins of life: why chirality is "biased" on Earth.
Chirality – from the Greek word for hand – is a property of individual molecules. If a molecule is chiral then we know that it has an enantiomer, a twin essentially and individual enantiomers are therefore often designated as either “right-” or "left-handed".
Many biologically active molecules are chiral, including naturally occurring sugars and amino acids (the building blocks of proteins) and are essential to life as we know it. Although most amino acids can exist in both left and right handed forms, living things are selective about which "handedness" of a molecule they use or produce. Life on Earth is made of left handed amino acids, almost exclusively, while all sugars in the metabolic pathways and in DNA and RNA are ‘right-handed’.
The use of only one handedness of any given molecule is known as homochirality and it is considered to be evolutionarily advantageous, however scientists do not yet understand how biology came to rely on one handedness and not the other, although it is widely believed that Earth life's "choice" of chirality was purely random.
"Homochirality is one of the most interesting properties of life as we know it," says Geoffrey Blake, professor of cosmochemistry and planetary sciences and professor of chemistry. "How did it come to be that all living things use one enantiomer of a particular amino acid, for example, over another? If we could run the tape of life again, would the same enantiomers be selected through a deterministic process, or is a random choice made that depends on a tiny imbalance of one handedness over the other? If there is life elsewhere in the universe, based on the biochemistry we know, will it use the same enantiomers?"
It is possible therefore that any carbon-based life forms elsewhere in the universe, could theoretically have opposite chirality. Nonetheless, chiral molecules have intriguingly been found in meteorites on Earth and comets in our Solar System, prompting the suggestion that radiation in the interstellar medium could have caused the selective destruction of one chirality of amino acids sequestered away in comet dust, leading to a selection bias that ultimately resulted in all life on Earth becoming homochiral.
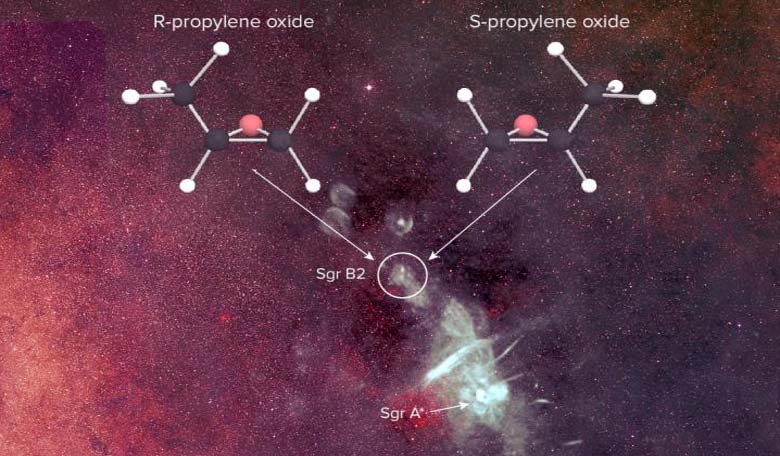
Now, with the discover of propylene oxide (CH3CHOCH2) found in an enormous star-forming cloud known as Sagittarius B2 (Sgr B2) by researchers primarily using the National Science Foundation's Green Bank Telescope (GBT) in West Virginia as part of the Prebiotic Interstellar Molecular Survey, this puzzle is one step closer to being solved.
“It's the first molecule detected in space that has the property of chirality, making it a pioneering leap forward in our understanding of how prebiotic molecules are made in space and the effects they may have on the origins of life," says Brandon Carroll, co-first author on the paper and a graduate student in Blake's group. "While the technique we used does not tell us about the abundance of each enantiomer, we expect this work to enable future observations that will let us understand a great deal more about chiral molecules, the origins of homochirality, and the origins of life in general."
Although propylene oxide is not utilised in living organisms, it is a useful molecule to study because it is relatively small compared to biomolecules such as amino acids and its presence in space is a signpost for the existence of other chiral molecules. "The next step is to detect an excess of one enantiomer over the other," says Brett McGuire, an NRAO Jansky Fellow and former member of the Blake lab, who shares first authorship on the work with Carroll. "By discovering a chiral molecule in space, we finally have a way to study where and how these molecules form before they find their way into meteorites and comets, and to understand the role they play in the origins of homochirality and life."